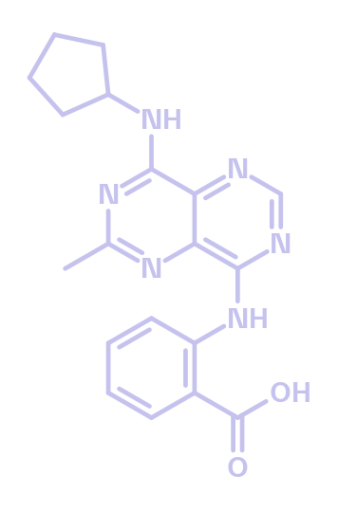
Multidrug resistant protein 4 (MRP4) inhibitor
BI-4926
BI-4926 is a potent small-molecule inhibitor of multidrug resistance protein 4 (MRP4/ABCC4). Based on its pharmacokinetic results in rat, this compound is suitable for in vivo and in vitro testing. It displays low double digit nanomolar activity in inhibiting cyclic AMP efflux from WI-38 cells and good blood-brain barrier penetration abilities. BI-5106 serves as a negative control.
More information
The human ATP-binding cassette transporter subfamily C (ABCC) consists of 12 integral membrane proteins (ABCC1-ABCC12). Nine of these (ABCC1–ABCC6, ABCC10–ABCC12) form the group of multidrug resistance proteins (MRP1–MRP9), which mediate the ATP-dependent export of organic anions from cells. Their extensive tissue distribution and plethora of substrate specificities render MRPs important factors to consider in the context of drug delivery.
MRP4 is an ATP-dependent efflux transporter for cyclic AMP (cAMP), cyclic GMP (cGMP), nucleotide analogs1,2, prostaglandins3,4, thromboxanes4, folic acid5, and many other amphiphilic substances. MRP4 is expressed in a number of tissues and cell types including brain capillary endothelial cells6, hepatocytes7, renal proximal tubule cells2, platelets8, and erythrocytes9. MRP4 overexpression in tumor cells is associated with resistance to a wide range of antineoplastic agents.
In the central nervous system, MRP4 is expressed in astrocytes, suggesting a role in lowering intracellular cGMP levels, and the release of cAMP and cGMP into the extracellular milieu. Extracellular cGMP has been shown to control intracellular pH in rat astrocytes, inhibit cellular responses of cerebellar neurons mediated by kainate receptors and protect against glutamate-induced toxicity6.
In hepatocytes, transport of bile salts by MRP4 was negligible in the absence of ATP or without S–methyl–glutathione7. These findings identify a novel pathway for the efflux of GSH across the basolateral hepatocyte membrane into blood where it may serve as an antioxidant and as a source of cysteine for other organs. Moreover, MRP4–mediated bile salt transport across the basolateral membrane may function as an overflow pathway during impaired bile salt secretion across the canalicular membrane into bile. In conclusion, MRP4 can mediate the efflux of GSH from hepatocytes into blood by cotransport with monoanionic bile salts7.
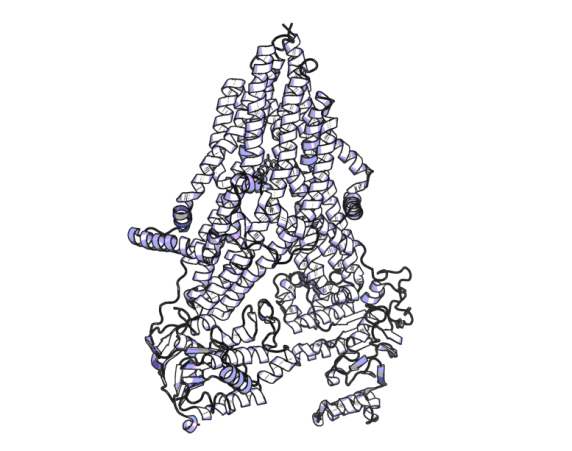
Structure of MRP4 in complex with one of its substrates (methotrexate) as resolved by cryo-EM (PDB ID: 8BWP)10.
Inhibition of MRP4 activities was measured in 2 assays: Firstly, a cellular cAMP export assay, and secondly a cAMP transport assay with membrane vesicles containing recombinant MRP4.
| Probe name / Negative control | BI-4926 | BI-5106 |
| MW [Da, free base]a | 364.4 | 436.5 |
| cAMP release from WI-38 cells (IC150) [nM]b | 25 | - |
| MRP4-mediated cAMP transport in membrane vesicles (IC150) [nM]b | 71 | >30,000 |
a The molecule is supplied in salt form; for the molecular weight of the salt, please refer to the vial label
b Cellular cAMP export assay:
WI-38 (human lung fibroblast cell line) cells were preincubated with the test compound at different concentrations and subsequently stimulated with isoprenaline in combination with a PDE4 inhibitor. cAMP released into culture supernatant was then quantified. In the cAMP transport assay, membrane vesicles prepared from insect cells (Hi-5) expressing recombinant human MRP4 were pre-incubated with test compound. The cAMP transport was initiated by adding a mixture of 3H-cAMP, ATP, and an ATP regenerating system (phosphocreatine, creatine kinase).
Culture medium, EMEM, Isoprenalin: 10 mM in EMEM
PDE4 inhibitor (e.g. roflumilast)
Assay procedure
1) Seed WI-38 VA13 (ATCC: CCL75.1) cells into 96-well cell culture plates 24 h before experiment (50,000 cells/well). To start the experiments, culture medium is removed, cells washed once with EMEM (without any additives).
2) Serial dilution of compounds: Maximal concentration 1.5 mg/ml in DMSO, 6 additional 5-fold serial dilutions in DMSO in a dilution plate (100x stock). Transfer 6 μl from the dilution plate into a deep-well plate containing 594 μl EMEM (without any additives). Transfer 180 μl to the cells. Incubate 30 min at 37°C.
3) Prepare stimulation mix: Stimulation mix containing a PDE4 inhibitor at appropriate concentration and isoprenalin (100 μM) is prepared with EMEM (without any additives). Add the mix (20 μl/well) to the cells. Incubate 30 min at 37°C.
4) Shake the plate at 600 rpm for 30 s. Transfer supernatant into a new plate.
5) Measure cAMP in supernatant with Catchpoint cAMP assay (Molecular Devices, R8089).
C Inhibitory potency of the compounds an MRP4: was measured using a filtration assay with 3H-cAMP:
A) Membran extraction, Solutions: Hypotonic buffer, 1 mM Tris, 1 mM Hepes, 0.1 mM EDTA, pH = 7.4 Tris/Sucrose-Puffer A (TS-A), 10 mM Tris, 250 mM Sucrose, pH = 7.4, Complete protease inhibitor cocktail tablets without EDTA (Roche diagnostics, 11 873 580 001)
Tris/Sucrose-Puffer A (TS-B), 10 mM Tris, 250 mM Sucrose, pH = 7.4
HEPES/KOH, 5 mM HEPES, adjust pH to 7.4 with KOH
38% Sucrose / HEPES, dissolve 38g Sucrose in HEPES/KOH, fill up to 100 ml
Cells:Hi5 insect cells, Medium: Insect Xpress (Cambrex # 12-730), Seed cells at 0.3 x 10E6 / ml 24 h before infect. Start baculo-virus infection with 5 ml virus stock. Harvest cells 72 h after start of infection by centrifugation at 3,000 rpm (SLA-3000) and 4 °C for 10 min.
Membrane extraction MRP4: Cell pellet (5x108 cells) was resuspended in 120 ml hypotonic buffer, stirred on ice for 90 min. Ultracentrifugation at 38,000rpm (TFT 50.38) and 4°C for 30 min. Pellet resuspended in 30 ml TS-A and pottered (30 strokes). Ultracentrifugation at 33,000rpm (TFT 50.38) and 4°C for 45 min. Pellet resuspended in 15 ml TS-B and pottered with (20 strokes). Aliquots frozen with liquid nitrogen and stored at -80°C.
B) Filtration assay
Material: Assay plates: Glass fiber type B filter plates (Millipore, MSFBN6B50), Compound dilution plates (Greiner, #651101), Scintillation fluid: Ultima Gold (PerkinElmer, 6013329)
Assay buffer: TS-B containing 0.1% BSA, 10x ATP, 247.8 mg ATP (Sigma, A8937), 181.4 mg Phosphocreatine (Sigma P1937), 203.3 mg MgCl2, 41.46 mg K2CO3, Dissolve in 4 ml TS-B, monitor pH (7.4), if necessary adjust with K2CO3 granules
Creatine kinase (Roche Diagnostics, 10736988001)
3H-cAMP: 2,8-3H-cAMP (Hartmann Analytic MT616, Biotrend ART-0163), 1 µCi/µl, cAMP: 10 mM Stock (3.29 mg/ml in TS-B)
Compounds: Stock 5 mg/ml in DMSO
Assay procedure: Assay materials are added into assay plates in sequence as follows: 1) compounds, 2) membrane mixture, 3) reaction mixture.
1) Serial dilution of compounds: Maximal concentration approximately 0.1 mM (dependent on molecular weight) in DMSO, 6 additional 5-fold serial dilutions in DMSO in a dilution plate (100x stock). Transfer 3 μl from the dilution plate into a plate containing 97 μl assay buffer (3.3x stock). Transfer 20 μl of the 3.3x stock into the assay plate.
2) For one assay plate, prepare the membrane mixture as follows: Dilute 4.4 mg membrane protein in assay buffer to a final volume of 2.2 ml. Add 20 μl / well of the membrane mixture to the assay plate.
3) For one assay plate, prepare the reaction mixture as follows: dilute 0.66 ml 10x ATP, 0.69 mg creatine kinase, 66 µl cAMP, and 55 µl 3H-cAMP in assay buffer to a final volume of 2.2 ml. Add 20 μl / well of the reaction mixture to the assay plate.
Transport reaction is initiated by the addition of the reaction mixture. After 60 min incubation at 37°C, 50 µl/well reaction mix is transferred into a filter plate with 200 µl/well pre-cold assay buffer. Plates are filtered with a vacuum manifold (Millipore, MSVMHTS00) and washed three times with assay buffer (pre-cooled with ice). Plates are dried at 37°C for 1 h in an incubator, 50 µl / well Ultima Gold is given to the plate. Radioactivity is quantified with Microbeta Trilux (PerkinElmer) using coincidence measurement.
Data analysis:
Each assay plate contains 6 high values (DMSO control) and 6 low values (50 µM BIBX2172BS). Data are expressed as percent control (average of the high values is set as 100%). Average of the low values (expressed as percent control) is used as bottom in the curve fitting process in Graphpad Prism program for the calculation of IC50 values.'
BI-4926 is highly permeable and binds strongly to plasma proteins. It has low risks for interaction with drug-metabolizing enzymes of the cytochrome P450 superfamily.
| Probe name / negative Control | BI-4926 | BI-5106 |
| logD @pH 2 / pH 7.4 / pH 11 | - / 1.9 / - | - |
| Solubility @ pH 7 [µg/ml] | 60 | <1 |
| Caco-2 permeability PAB @ 10µM [10-6 cm/s] | 81 | 0.2 |
| Caco-2 efflux ratio | 1.1 | 50.7 |
| Microsomal stability (human/mouse/rat) [% QH] | 80 / <46 / 62 | <23 / - / <22 |
| Hepatocyte stability (human/mouse/rat) [% QH] | 86 / - / 67 | - |
| Plasma protein binding (rat) [%] | 99.7 | 93.4 |
| CYP 3A4 (IC50) [µM] | >50 | >50 |
| CYP 2C8 (IC50) [µM] | 19.6 | 49.2 |
| CYP 2C9 (IC50) [µM] | 3.4 | >50 |
| CYP 2C19 (IC50) [µM] | >50 | >50 |
| CYP 2D6 (IC50) [µM] | >50 | >50 |
At a p.o. dose of 30 mg/kg (Natrosol suspension) in rat, BI-4926 achieved a plasma exposure of 4.3 µM at 2 h post dosing. Exposure in CSF was similar to the unbound exposure in plasma indicating good CNS penetration of the compound.
| BI-4926 | Rata |
| Clearance [% QH] | 34 |
| Mean residence time after i.v. dose [h] | 0.3 |
| tmax [h] | 0.62 |
| Cmax [nM] | 2,830 |
| F [%] | 58 |
| Vss [L/kg] | 0.4 |
a i.v. dose: 0.4 mg/kg, p.o. dose: 4.0 mg/kg
In vivo DMPK parameters
BI-5106, a structurally close analog can be used as a negative control.
BI-5106 which serves as a negative control.
BI-4926 was tested against the close homologs MRP2 and MRP5 and showed 57-fold and 429-fold selectivity, respectively. In Cerep selectivity screen against 79 targets, BI-4926 inhibited 2 GPCRs by more than 50% at a concentration of 10 µM: Prostaglandin E2 receptor 2 and Adenosine A3 receptor.
The negative control BI-5106 showed in 3 out of 44 targets inhibition with more than 50% @ 10µM (COX-1, PDE4D2 and PDE3A).
| SELECTIVITY DATA AVILABLE | BI-4926 | BI-5106 |
SafetyScreen44™ with kind support of  | Yes | Yes |
| Invitrogen® | Yes | No |
| DiscoverX® | No | No |
| Dundee | No | No |
Download selectivity data:
BI-4926_selectivityData.xlsx
BI-5106_selectivityData.xlsx
BI-4926 is a potent and selective inhibitor of multidrug resistance protein 4 (MRP4), suitable for in vitro as well as in vivo studies. BI-5106, a structurally close analog, can be used as a negative control.
The human multidrug resistance protein MRP4 functions as a prostaglandin efflux transporter and is inhibited by nonsteroidal antiinflammatory drugs
Reid G., Wielinga P., Zelcer N., van der Heijden I., Kuil A., Haas M. de, Wijnholds J., Borst P.
Proc Natl Acad Sci U S A 2003, 100(16), 9244–9249.
Analysis of methotrexate and folate transport by multidrug resistance protein 4 (ABCC4): Mrp4 is a component of the methotrexate efflux system
Chen Z.‑S., Lee K., Walther S., Raftogianis R. B., Kuwano M., Zeng H., Kruh G. D.
Cancer Res 2002, 62(11), 3144–3150.
The nucleotide transporter MRP4 (ABCC4) is highly expressed in human platelets and present in dense granules, indicating a role in mediator storage
Jedlitschky G., Tirschmann K., Lubenow L. E., Nieuwenhuis H. K., Akkerman J. W. N., Greinacher A., Kroemer H. K.
Blood 2004, 104(12), 3603–3610.