Voltage-gated Na+ channel 1.1 activator
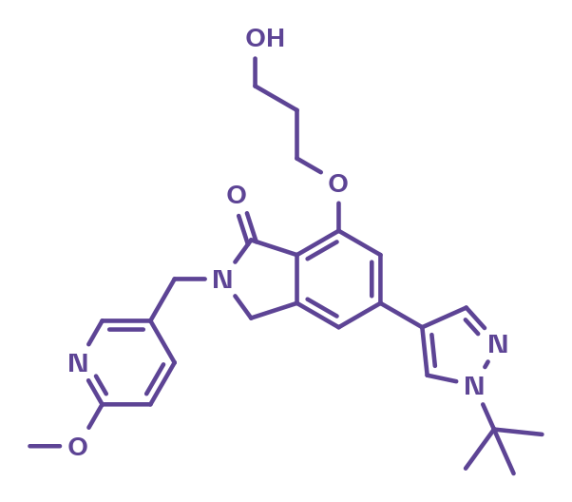
BI-7150
BI-7150 is a potent small-molecule activator of voltage-gated sodium channel Nav1.1 that can be used for in vitro studies. The compound is representative of a structural class which is different from other known Nav1.1 activators1. BI-7150 displays good aqueous solubility, as well as reduced protein binding.
More information
The voltage gated sodium channel Nav1.1 comprises a single pore-forming α-subunit and two β-subunits. Nav1.1 is primarily expressed in the central nervous system, where it is involved in neuronal membrane depolarization and thereby in action potential firing. Within the CNS, Nav1.1 is predominantly expressed in the GABAergic parvalbumin positive fast spiking interneurons. This sub-population of interneurons regulates the excitatory state of the glutamatergic pyramidal neurons and thereby is involved in controlling the excitatory / inhibitory balance (E/I balance)1-8. Inappropriate function of fast-spiking interneurons can result in disinhibition of pyramidal cells and network desynchronization, without detectable changes in excitatory pyramidal neurons2-3. The resulting imbalance between excitation and inhibition is thought to contribute to hyperexcitability and seizures.
Nav1.1. is encoded by the SCN1A gene, for which up to 900 missence, nonsense and truncation mutations have been identified. Such mutations can lead to loss of function or gain of function phenotypes4-5. While the pathogenic mechanism of such mutations is not yet understood, a clear genetic link has been established between SCN1A mutations and Dravet syndrome, also known as the severe myoclonic epilepsy of infancy3,6.
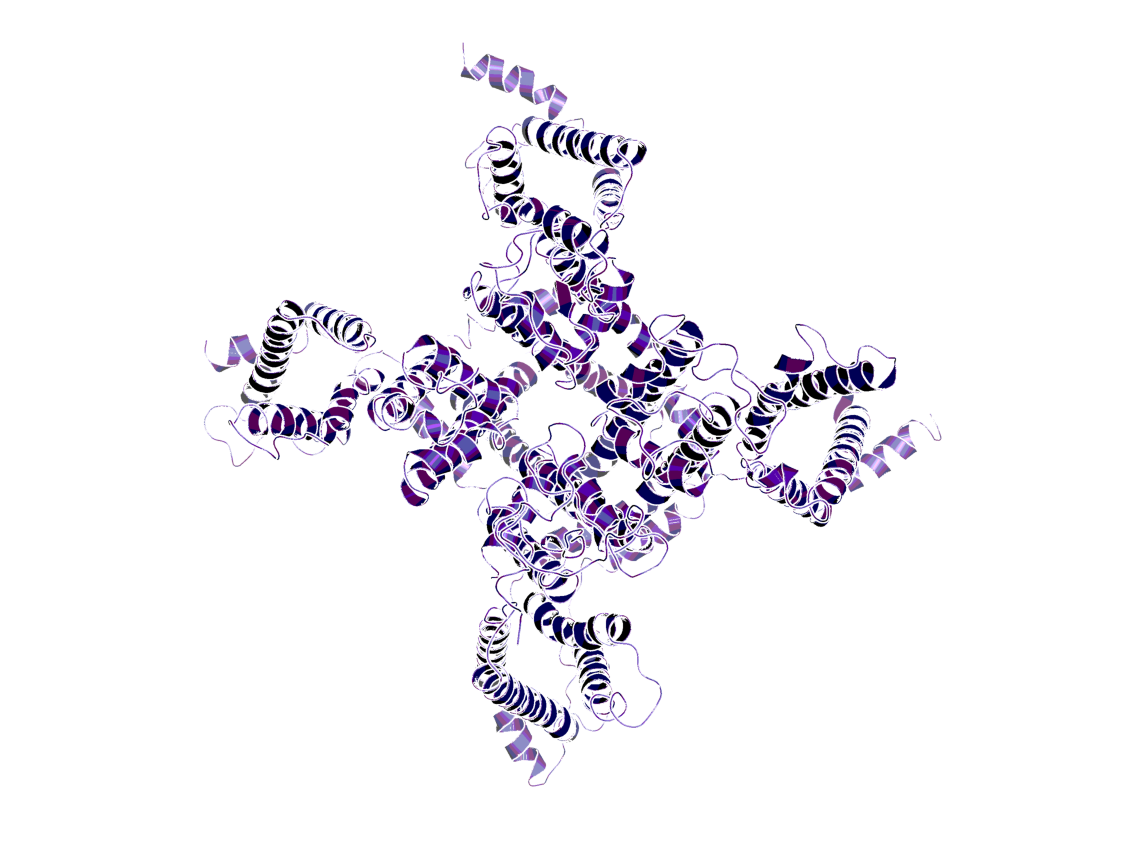
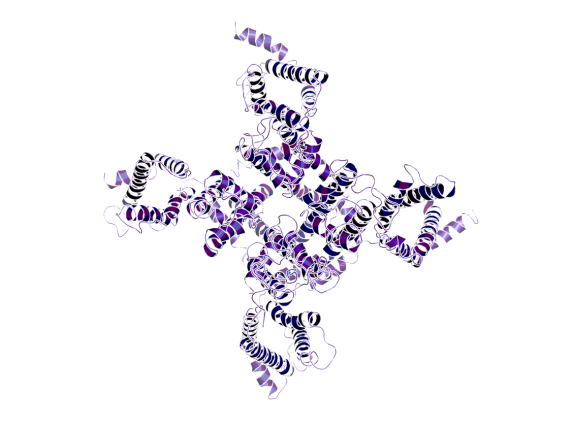
Model of the voltage-gated sodium channel Nav1.1 (PDB code: 7DTD)2.
Potentiation of Nav1.1 was measured via an automated sychropatch electrophysiology assay and based on sodium currents caused by the opening (activation) of the voltage-gated sodium channel recombinantly expressed in HEK 293 cells. Channel activation was evoked by the depolarization of the cell, which changes the channel into its open state. The transfer of sodium ions in the open state can be recorded using the patch clamp technique.
Compounds were tested in an 11-point concentration curve in quadruplicate starting at 30µM and using a 1:3 dilution factor with a constant DMSO concentration of 0.3%. Once cells were plated and compounds had been added, a holding potential of –100 mV was applied during the first 50 ms to measure baseline recordings. This was followed by a depolarization to 0 mV for 50 ms. Over the course of these 50 ms, peak current, TAU (time constant of inactivation), AUC (area under the curve), and residual current were recorded and analyzed. Subsequent to this depolarization, the holding potential of –100 mV was reapplied for 50 ms prior to the next of a total of 46 sweeps.
| Probe name / Negative control | BI-7150 | BI-7283 |
| MW [Da, free base]a | 450.5 | 475.6 |
| Nav 1.1 TAU (EC150) [nM]b | 119 | 14,500 |
| Nav 1.1 AUC (EC150) [nM]c | 45 | 4,440 |
a The molecule is supplied in salt form. For the molecular weight of the salt form, please refer to the vial label
b EC150 is defined as the compound concentration necessary to increase the Nav1.1 ion throughput over 50 ms by 50% relative to stimulation in the presence of vehicle. The biological activity of compounds is determined by the following methods:
1) Syncropatch 384PE platform and software
The activity of voltage-dependent sodium (Nav) channels and cpd-dependent modulation of Nav channel activity were recorded with the automated patch clamp platform Syncropatch 384PE utilizing NPC-384 resistance chips (384-well format). Data acquisition was controlled with PatchControl384 software, and export and analysis were performed with DataControl384 software (all Nanion Technologies, Munich, Germany) and customized analysis software.
2) Buffers used in the experiments
The following extracellular solutions were utilized: Cell catch buffer containing (in mM) 140 NaCl, 4 KCl, 2 CaCl2, 1 MgCl2, 10 Glucose, 10 HEPES (pH adjusted to 7.4 with NaOH). Seal enhancer buffer containing (in mM) 80 NaCl, 3 KCl, 35 CaCl2, 10 MgCl2,10 HEPES (pH adjusted to 7.4 with NaOH; cell catch buffer). Washing/run buffer containing (in mM) 140 NaCl, 4 KCl, 5 CaCl2, 1 MgCl2, 10 Glucose, 10 HEPES (pH adjusted to 7.4 with NaOH; cell catch buffer). Washing/run buffer containing (in mM) 140 NaCl, 4 KCl, 5 CaCl2, 1 MgCl2, 10 Glucose, 10 HEPES (pH adjusted to 7.4 with NaOH; cell catch buffer).
The intracellular solution was prepared by mixing to two buffers (90 vol% of buffer I with 10% of buffer II). Buffer I contained (in mM) 140 CsF, 10 NaCl, 1 EGTA, 10 HEPES and Buffer II contained (in mM) 20 EDTA, 20 MgCl2. After addition of 1 mM Na2ATP, the pH of the intracellular buffer was adjusted to 7.2 (with CsOH).
3) Cell types
Experiments were performed utilizing HEK293 cells stably expressing SCN1A (NM_006920 (var 2); pD3.2 vector) and SCN1B (NM_001037.4; pD6.2 vector) encoding human Nav channel α subunit and β1 subunit, respectively (termed HEK-Nav1.1 cells; from SB DrugDiscovery).
4) Compound plate preparation:
Compound plates were prepared by Compound Logistic in Axygen deep well plates 384 (Thermo Fisher Scientific, Waltham, US), compound wells contained a volume of 300 nL. Each compound titration was prepared in quadruplicates in 100% DMSO. Starting from the highest concentration of 10 mM, compounds were in diluted 1:3 on the plate to create a 11-point concentration-response curve. Compounds were then diluted by adding 50µl/well of run buffer containing a 0.1% Pluronic F-127 solution. An extra 1:2 dilution was carried out by the Syncropatch during the experiments, reaching a final top concentration of 30µM and 0.3% DMSO. Controls were manually prepared freshly on the day of the experiment. 0.3% DMSO was used as negative control and ATX II (30 nM in 0.3% DMSO) served as positive control.
5) Voltage protocol, data sampling, analysis, and quality control
From a holding potential of -100 mV, Nav channels were activated by depolarizing voltage steps to 0 mV for 50 ms every 20 s. A total of 24 baseline sweeps and 17 sweeps after compound application were recorded, leak subtraction was applied in between sweeps. The following parameters were analyzed: baseline (leak current), peak inward current, AUC, residual current after channel inactivation, TAU, R2, seal resistance, cell capacitance, Rs (series resistance). Recordings were only analyzed, if current amplitude before application of cpd was >-200 pA, and if Rs was 30 MΩ throughout recordings. Recordings were discarded, if the following quality control criteria were not met: Residual current > 50 pA, baseline current <-100 pA.
c EC150 is defined as the compound concentration necessary to increase the Nav1.1 ion throughput over 50 ms by 50% relative to stimulation in the presence of vehicle.
BI-7150 shows good aqueous solubility and permeability as well as acceptable metabolic stability across human, rat, and mouse hepatocytes. The compound’s protein binding in human and mouse plasma appears to be high, but still with reasonable fraction unbound, in line with a measured logD of 2.9.
| Probe name / negative Control | BI-7150 | BI-7283 |
| logD @pH 2 / pH 7.4 / pH 11 | 2.2 / 2.9 / n.a. | 1.5 / n.a. / 3.4 |
| Solubility @ pH 7 [µg/ml] | 222.2 | 73 |
| MDCK permeability PAB @ 1µM [10-6 cm/s] | 19 | n.a. |
| MDCK efflux ratio | 5.3 | n.a. |
| Microsomal stability (human/mouse/rat) [% QH] | <33 / n.a. / 25 | 70 / 65 / 51 |
| Hepatocyte stability (human/mouse/rat) [% QH] | 40 / 73 / 36 | n.a. |
| Plasma protein binding (human/mouse) [%] | 90.7 / 94.4 | n.a. |
| CYP 3A4 (IC50) [µM] | >50 | n.a. |
| CYP 2C8 (IC50) [µM] | 24.1 | n.a. |
| CYP 2C9 (IC50) [µM] | >50 | n.a. |
| CYP 2C19 (IC50) [µM] | >50 | n.a. |
| CYP 2D6 (IC50) [µM] | >50 | n.a. |
Despite its good aqueous solubility, brain-blood barrier penetration and bioavailability, use of BI-7150 in vivo is not recommended due to adverse effects observed in murine models.
BI-7283 was designed as a structurally close analogue of BI-7150 and can be used as a negative control.
BI-7283 which serves as a negative control.
BI-7150 was tested on 44 targets in a selectivity panel and showed ≥1,000fold selectivity for 42 targets (≤ 50%inhibition @ 10 µM). In two assays (5HT2H/H and LCK_CE) the compound showed inhibition between 55% and 98% @ 10µM.
The negative control BI-7283 showed in 9 out of 44 targets inhibition with more than 50% @ 10 µM (D2SH_AGON, NEUP/H, M3/H, MU/H, ACE/HU, M2/H, M1/H, Na+/SITE2, HERG_DOFETILIDE).
| SELECTIVITY DATA AVILABLE | BI-7150 | BI-7283 |
SafetyScreen44™ with kind support of  | Yes | Yes |
| Invitrogen® | No | No |
| DiscoverX® | No | No |
| Dundee | No | No |
Download selectivity data:
BI-7150_selectivityData.xlsx
BI-7283_selectivityData.xlsx
Nav1.1 activator 1, HY-1264291
AA432792
BI-7150 is a potent and selective activator of the voltage-gated sodium channel Nav1.1. BI-7283 is available as negative control.
A small molecule activator of Nav 1.1 channels increases fast-spiking interneuron excitability and GABAergic transmission in vitro and has anti-convulsive effects in vivo
Frederiksen K., Lu D., Yang J., Jensen H. S., Bastlund J. F., Larsen P. H., Liu H., Crestey F., Dekermendjian K., Badolo L., Laursen M., Hougaard C., Yang C., Svenstrup N., Grunnet M.
Eur J Neurosci 2017, 46(3), 1887–1896.