TRPC4/5 inhibitor
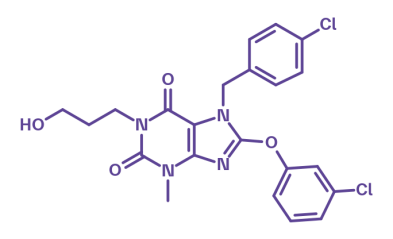
HC-070
HC-070 is a potent, selective inhibitor of TRPC4/5 channels1, key regulators of neuronal excitability. By reducing calcium influx, HC-070 restores neuronal balance2,3 and shows robust anxiolytic and antidepressant-like effects in vivo1. Its defined pharmacological profile makes it a valuable tool for exploring TRPC4/5 biology in the CNS and peripheral tissues, where dysregulated channel activity may impact physiological processes.
More information
TRPC4 and TRPC5 belong to the transient receptor potential canonical family of non-selective cation channels. They form tetrameric complexes permeable to calcium and sodium and are widely expressed in brain regions such as the amygdala and prefrontal cortex. Activation of these channels via G-protein-coupled receptor signalling leads to sustained calcium entry and increased neuronal excitability.
Preclinical data indicate that TRCP4/5 channels may be involved in a wide range of processes—from innate fear, anxiety, and pain signaling to cardiovascular processes, such as vascular tone and cardiac remodeling, and emerging roles beyond. Yet their precise physiological roles remain unclear due to the limited availability of selective research tools.
Hence, pharmacological inhibition of TRPC4/5 may provide a new promising approach to normalize excitability within affected neural circuits and explore new roles of TRPC4/5 signaling4.

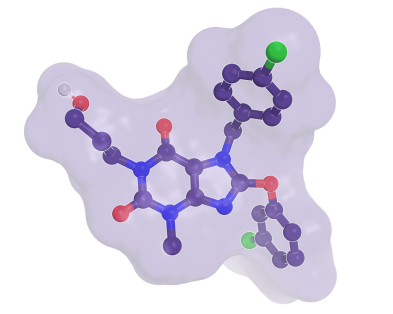
Structure of the complex of Pico 145, an inhibitor closely related to HC-070, bound to a TRPC1/4 heterotetramer (PDB code: 8WPM5). Four binding sites of the tetrameric structure are occupied with the inhibitor.
HC-070 displays potent dual activity on both TRPC4 and TRPC5 channels with an IC50 ≤ 0.5 nM in human and rodent patch clamp assays1.
| Probe name | HC-070 |
| MW [Da] | 475.32 |
| TRPC5 La3+ (IC50) (human/mouse/rat) [nM]a | 0.5/0.5/0.3 |
| TRPC5 with20 µM carbachol (IC50) (human/mouse [nM]a | 2.0/1.8 |
| TRPC4 with 10µM carbachol (IC50) (human/mouse) [nM]a | 1.8/0.5 |
a See ref. 1
HC‑070 shows poor solubility and a low fraction unbound. Its low MDCK efflux ratio indicates favorable brain penetration, making it well suited for CNS‑focused studies.
| Probe name | HC-070 |
| logP | 5.3 |
| Solubility @ pH 6.8 [µg/ml] | <1.0 |
| Caco-2 permeability @ pH 7.4 [*10-6 cm/s] | 134.0 |
| Caco-2 efflux ratio | 0.8 |
| MDCK permeability Papp AB @ 1µM [10-6 cm/s] | 28.1 |
| MDCK efflux ratio | 0.6 |
| Hepatocyte stability (human/mouse/rat) [% QH] | 76/82/78 |
| Plasma Protein Binding (mouse) [%] | 99.6 |
| hERG (IC50) [µM] | 2 |
| CYP 1A2 (IC50) [µM] | >50 |
| CYP 3A4 (IC50) [µM] | >50 |
| CYP 2C8 (IC50) [µM] | 16 |
| CYP 2C9 (IC50) [µM] | 8 |
| CYP 2C19 (IC50) [µM] | 27 |
| CYP 2D6 (IC50) [µM] | 22 |
| CYP 2B6 (IC50) [µM] | >50 |
HC‑070 shows effective CNS penetration together with favourable oral bioavailability in mice.
| HC-070 | Mouse |
| Clearance [% QH]a | 24 |
| Mean residence time after i.v. dose [h]a | 1.9 |
| tmax [h]b | 1 |
| Cmax [nM]b | 937 |
| F [%]b | 13 |
| Vss [l/kg]a | 1.5 |
a i.v. dose 1 [mg/kg]
b p.o. dose 10 [mg/kg]
HC‑070 demonstrates significant efficacy in the Chronic Social Defeat (CSD) test, a model in which mice are exposed to prolonged social stress that enhances fear learning and memory. These results indicate that TRPC4/5 inhibition acts to reverse the CSD stress effect of increased consolidation/expression of the fear memory of the discrete CS and the context. The time course of the behavioural effects further suggests that the primary action of HC‑070 is to facilitate the extinction of these fear memories1.

Effects of HC-070 on CSD-induced fear hyper-reactivity1.
HC-070 reduces the increased capacity for fear memory in mice exposed to chronic social stress on days 1–15. (A) Day 16: Without drug administration, when placed in a relatively unfamiliar arena (context) without tone or electroshock, mice that had been exposed to chronic social defeat (CSD) tended to show more freezing—a fear behavior—than did control mice (CON). (B) Day 17: Without drug administration, mice were placed back in the same context and exposed to 6 pairings of a 20 s tone conditioned stimulus (CS) that announced a 2 s electroshock unconditioned stimulus (US). CSD mice acquired more freezing to the CS than did CON mice. The left panel presents the fear learning curve using the average freezing during CS-US trials 1–2, 3–4 and 5–6, and the right panel presents the average freezing across all six CS-US trials. Immediately after this CS-US conditioning session, CSD and CON mice received either vehicle or 1 mg/kg HC-070 orally. (C) Day 18: Mice received vehicle or 1 mg/kg HC-070 orally and 1 hour later were placed in the same context in which CS-US conditioning took place the previous day, and a 21-min test of context fear memory was conducted, i.e. in the absence of the CS (and US). The left panel presents the context-fear expression curve using the average freezing per 3-min block. The right panel presents the average context-fear expression across all 21 min. HC-070 significantly reduced the context fear memory in CSD mice, as indicated by these mice showing freezing levels similar to those of CON mice and lower than those of CSD-VEH mice. (D) Day 18: Immediately after the context fear memory test, the tone-CS fear memory test was conducted, comprising 12 trials of 30-s CS separated by 90-s inter-trial intervals. The fear expression curve to the CS is presented using the average freezing per pair of consecutive CS trials. HC-070 tended to reduce the CS fear memory in CSD mice, as indicated by the increased rate at which their freezing level during the CS attenuated compared with CSD-VEH mice, i.e. faster extinction learning. (E) Day 18: In the tone CS memory test presented in (D), freezing was also measured in the inter-trial intervals (ITIs) between CS presentations. The left panel presents the fear expression curve in ITIs between CSs using the average freezing per pair of consecutive ITIs. The right panel presents the average freezing across all 12 ITIs. Here also, HC-070 reduced fear memory in CSD mice, as indicated by the increased rate at which their freezing level during the ITIs attenuated compared with CSD-VEH mice, i.e. faster extinction learning.
HC-070 inhibited 8 out of 44 tested targets in the SafetyScreen44TM by greater than 50% @ 10µM (Na+/SITE2/R, KAPPA(KOP), HERG_DOFE, Delta2/H, 5HT2AH_Ago, PDE4D2, NEUP/H, 5HT2B/H AG).
| SELECTIVITY DATA AVAILABLE | HC-070 |
SafetyScreen44™ with kind support of  | Yes |
Download selectivity data:
HC-070_selectivityData.xlsx
HC-070, a potent TRPC4/5 inhibitor, reduces calcium influx, modulates neuronal excitability, and is a tool for studying TRPC4/5 in vivo.
Treatment with HC-070, a potent inhibitor of TRPC4 and TRPC5, leads to anxiolytic and antidepressant effects in mice
Just S., Chenard B. L., Ceci A., Strassmaier T., Chong J. A., Blair N. T., Gallaschun R. J., Del Camino D., Cantin S., D'Amours M., Eickmeier C., Fanger C. M., Hecker C., Hessler D. P., Hengerer B., Kroker K. S., Malekiani S., Mihalek R., McLaughlin J., Rast G., Witek J., Sauer A., Pryce C. R., Moran M. M.
PLoS One 2018, 13(1), e0191225.
Inhibition of Canonical Transient Receptor Potential Channels 4/5 with Highly Selective and Potent Small-Molecule HC-070 Alleviates Mechanical Hypersensitivity in Rat Models of Visceral and Neuropathic
Jalava N., Kaskinoro J., Chapman H., Morales M., Metsänkylä H., Heinonen S.‑M., Koivisto A.‑P.
Int J Mol Sci 2023, 24(4).
Picomolar, selective, and subtype-specific small-molecule inhibition of TRPC1/4/5 channels
Rubaiy H. N., Ludlow M. J., Henrot M., Gaunt H. J., Miteva K., Cheung S. Y., Tanahashi Y., Hamzah N., Musialowski K. E., Blythe N. M., Appleby H. L., Bailey M. A., McKeown L., Taylor R., Foster R., Waldmann H., Nussbaumer P., Christmann M., Bon R. S., Muraki K., Beech D. J.
J Biol Chem 2017, 292(20), 8158–8173.
When you plan a publication, please use the following acknowledgement:
HC-070 was kindly provided by Boehringer Ingelheim via its open innovation platform opnMe, available at https://www.opnme.com.