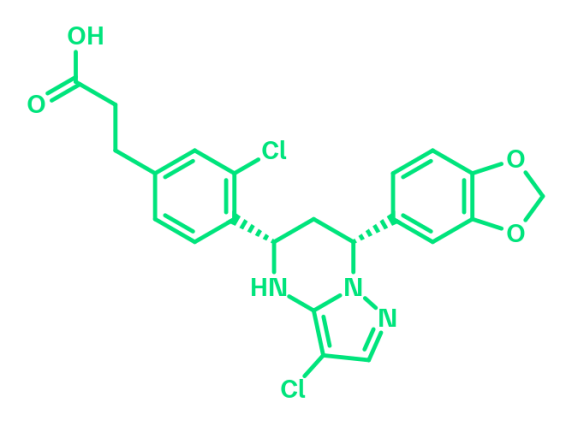
BCL6 degrader
BI-1136
With BI-1136, we share a selective, orally bioavailable BCL6 degrader with high potency. The molecule is suitable for in vivo testing in rodents. Mechanistically, the molecule acts as a BCL6 specific protein degrader and displays a strong induction of expression of BCL6-repressed genes. Having previously been offered as a molecule for collaboration, we now share BI-1136 with the entire research community.
More information
The transcriptional regulator BCL6 represses genes required for the differentiation of B-cells in germinal centers (GC)2. Errors in the GC reaction can give rise to mutated B cells that maintain an elevated proliferation and fail to differentiate, contributing to the genesis of diffuse large B-cell lymphoma (DLBCL)3. BCL6 is an oncogenic driver for DLBCL4,5,6,7 and its expression is frequently elevated by mutations in DLBCL. However, despite significant research efforts, the clinical relevance of targeting BCL6 in DLBCL remains to be proven.
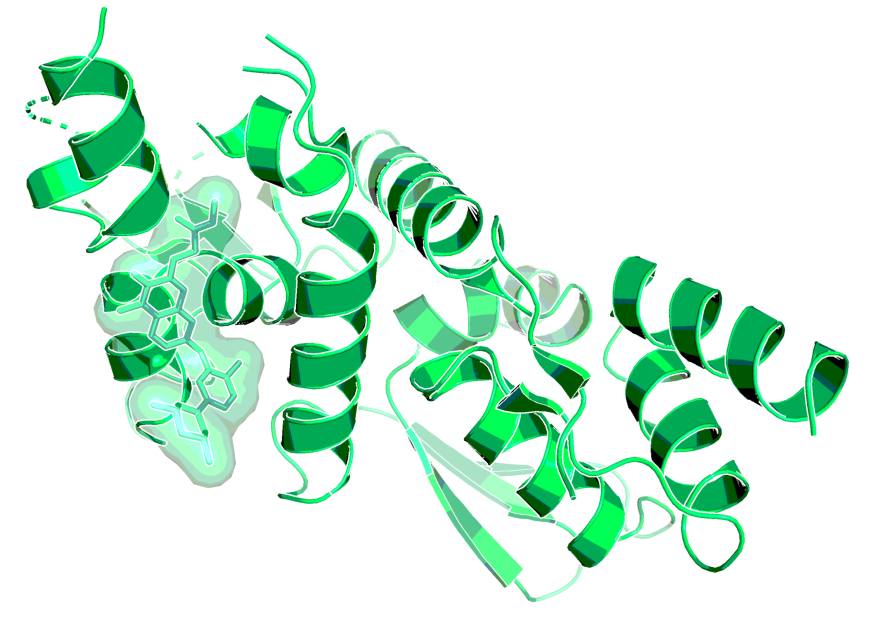
BCL6-BTB dimer with BI-3802, as observed by X-ray1. BI-3802 binds at the interface of two monomers (monomers are shown in green and grey).
BI-1136 is an unprecedented molecule that potently inhibits the interaction of the BTB/POZ domain of BCL6 with several co-repressors in vitro (IC50 ≤ 50 nM). In a cellular context, the BCL6 degrader inhibits the BCL6::co-repressor complex formation with an IC50 of 210 nM. Moreover, our BCL6 degrader was found to be a potent and efficacious degrader of the BCL6 protein in mouse and human DLBCL cell lines (DC50 = 63 nM in SU-DHL-4 cells) as well as in other BCL6-expressing cells tested (macrophages, NSCLC, Burkitt and breast cancer cell lines). It induces BCL6 target genes in SU-DHL-4 cells with an EC50 of 60-200 nM. Interestingly we found that the BCL6 degrader BI-1136 displays significantly stronger induction of expression of BCL6-repressed genes than compounds that merely inhibit co-repressor interactions. The BCL6 degrader is mouse cross reactive and highly selective for BCL6.
| PROBE NAME / NEGATIVE CONTROL | BI-1136 | BI-1135 |
| MW [Da, free base]a | 460.3 | 460.3 |
| BCL6::BCOR ULight TR-FRET (IC50) [nM]b | 44 | 17,625 |
| BCL6::NCOR LUMIER (IC50) [nM]b | 210 | >10,000 |
| BCL6 protein degradation (DC50) [nM]b, c | 63 | N/A |
a For the salt form you will get, please refer to the label on the vial and for the molecular weight of the salt, please refer to the FAQs
bFor assay conditions see ref. 1
c In SU-DHL-4 cells
BI-1136 has acceptable solubility in water at neutral pH, high permeability in Caco-2 and MDCK assays and medium plasma protein binding and stability in mouse liver microsomes.
| PROBE NAME / NEGATIVE CONTROL | BI-1136 | BI-1135 |
logP @ pH 2.0 | |
|
logD @ pH 2.0 | |
|
| Solubility @ pH 6.8 [µg/mL] | >93 | 90 |
| Caco-2 permeability AB @ pH 7.4 [*10-6 cm/s] | 19.8 | 23 |
| Caco-2 efflux ratio | 0.6 | 1.1 |
| MDCK permeability PappAB @ 1µM [10-6 cm/s] | 5.9 | 6.2 |
| MDCK efflux ratio | 2.4 | 2.6 |
| Microsomal stability (human/mouse/rat) [% QH] | 53 /32 / <23 | 43 / 51 / 43 |
| Hepatocyte stability (human/mouse/rat) [% QH] | 18 / 23 / 8 | 11 / 35 / 22 |
| Plasma Protein Binding (human/mouse/rat) [%] | 99.2 / 96.8 / 97.9 | 99.8 / 99.4 / >99.6 |
| hERG [inh. % @ 10 µM] | 17.4 | N/A |
| CYP 3A4 (IC50) [µM] | 13 | >50 |
| CYP 2C8 (IC50) [µM] | 5 | 12.5 |
| CYP 2C9 (IC50) [µM] | >50 | >50 |
| CYP 2C19 (IC50) [µM] | >50 | >50 |
| CYP 2D6 (IC50) [µM] | >50 | >50 |
The BCL6 degrader shows pharmacokinetic (PK) properties that are suitable for in vivo testing in several animal species and is well tolerated.
| BI-1136 | MOUSE | RAT |
| Clearance [% QH]a | 78 | 27 |
| Mean residence time after i.v. dose [h]a | 0.44 | 1.5 |
| tmax [h]b | 0.25 | 1.5 |
| Cmax [nM/µmol/kg]b | 59 | 155 |
| F [%]b | 27 | 56 |
| Vss [L/kg]a | 1.8 | 1.7 |
a i.v. dose: 2 mg/kg
b p.o. dose: 20 mg/kg
The PK properties of BI-1136 in several animal species render it suitable for once or twice daily oral dosing in acute or sub-chronic in vivo experiments, resulting in significant, but not complete degradation of BCL6 in SU-DHL-4 xenografts.
We encourage orders of BI-1135 as a negative control which is the inactive enantiomer of BI-1136 and displays a differentiated pharmacological profile.
BI-1135 which serves as a negative control
BI-1136 shows high selectivity at 10 µM concentration versus a panel of 44 receptors (no binding), Kinase panel (38 kinases, no hit at 10 µM). The compound is well tolerated in mice in doses up to 1 g/kg daily. The negative control BI-1135 showed inhibition with more than 50% @10 µM in 1 out of 44 targets. It showed binding in the endothelin (ETA/H) assay with an inhibition of 54% @10 µM.
| SELECTIVITY DATA AVAILABLE | BI-1136 | BI-1135 |
SafetyScreen44™ with kind support of  | Yes | Yes |
| Invitrogen® | Yes | No |
| DiscoverX® | No | No |
| Dundee | No | No |
BI-1136 is complementary to BI-3802, a potent BCL6 degrader with in vitro activity. For more information see references 8,9,10,11.
BI-1136 is an unprecedented, potent and selective orally bioavailable BCL6 degrader with rodent in vivo activity. A negative control (BI-1135) is available.
2D structure formats available
BCL6 degrader | BI-1136.smiles
Negative control | BI-1135.png
Negative control | BI-1135.smiles
Negative control | BI-1135.sdf
BI-1136 and its negative control BI-1135 have been designed as part of a collaboration with Forma Therapeutics. Forma is now a fully owned subsidiary of Novo Nordisk A/S.
Chemically induced degradation of the oncogenic transcription factor BCL6
Kerres N., Steurer S., Schlager S., Bader G., Berger H., Caligiuri M., Dank C., Engen J. R., Ettmayer P., Fischerauer B., Flotzinger G., Gerlach D., Gerstberger T., Gmaschitz T., Greb P., Han B., Heyes E., Iacob R. E., Kessler D., Kölle H., Lamarre L., Lancia D. R., Lucas S., Mayer M., Mayr K., Mischerikow N., Mück K., Peinsipp C., Petermann O., Reiser U., Rudolph D., Rumpel K., Salomon C., Scharn D., Schnitzer R., Schrenk A., Schweifer N., Thompson D., Traxler E., Varecka R., Voss T., Weiss-Puxbaum A., Winkler S., Zheng X., Zoephel A., Kraut N., McConnell D., Pearson M., Koegl M.
Cell Rep. 2017, 20, 2860-2875.
Integrated biochemical and computational approach identifies BCL6 direct target genes controlling multiple pathways in normal germinal center B cells
Basso K., Saito M., Sumazin P., Margolin A. A., Wang K., Lim W. K., Kitagawa Y., Schneider C., Alvarez M. J., Califano A., Dalla-Favera R.
Blood 2010, 115, 975-984.
Rationally designed BCL6 inhibitors target activated B cell diffuse large B cell lymphoma
Cardenas M. G., Yu W., Beguelin W., Teater M. R., Geng H., Goldstein R. L., Oswald E., Hatzi K., Yang S. N., Cohen J., Shaknovich R., Vanommeslaeghe K., Cheng H., Liang D., Cho H. J., Abbott J., Tam W., Du W., Leonard J. P., Elemento O., Cerchietti L., Cierpicki T., Xue F., MacKerell A. D. Jr., Melnick A. M.
J. Clin. Invest. 2016, 126, 3351-3362.
Small-molecule-induced polymerization triggers degradation of BCL6
Słabicki M., Yoon H., Koeppel J., Nitsch L., Roy Burman S. S., Di Genua C., Donovan K. A., Sperling A. S., Hunkeler M., Tsai J. M., Sharma R., Guirguis A., Zou C., Chudasama P., Gasser J. A., Miller P. G., Scholl C., Fröhling S., Nowak R. P., Fischer E. S., Ebert B. L.
Nature 2020, 588, 164–168.
Frequent mutations of FBXO11 highlight BCL6 as a therapeutic target in Burkitt lymphoma
Pighi C., Cheong T.C., Compagno M., Patrucco E., Arigoni M., Olivero M., Wang Q., Lopez C., Bernhart S.H., Grande B.M., Poggio T., Langellotto F., Bonello L., Dall'Olio R., Martinez-Martin S., Molinaro L., Francia di Celle P., Whitfield J.R., Soucek L., Voena C., Calogero R.A., Morin R.D., Staudt L.M., Siebert R., Zamo A., Chiarle R.
Blood Adv 2021, 5(23):5239-5257.
When you plan a publication, please use the following acknowledgement:
BI-1136 was kindly provided by Boehringer Ingelheim via its open innovation platform opnMe, available at https://www.opnme.com.