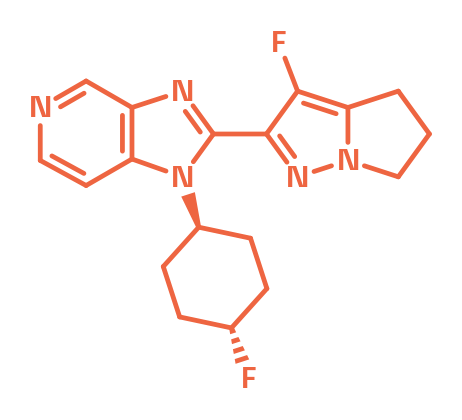
mGluR2 Positive Allosteric Modulator
BI-4737
mGlu2 receptors regulate synaptic glutamate release in the brain. Their activation can normalize over-activated circuits in the cortico-limbic system. BI-4737 is a mGluR2 positive allosteric modulator designed together with Evotec, that has shown to be potent in vitro and in vivo. BI-4576 serves as a negative control.
More information
mGlu2 receptors function in the brain as auto-receptors at the pre-synaptic terminals to down-regulate synaptic glutamate release. mGluR2 PAMs will enhance this inhibitory feedback mechanism at overexcited nerve terminals to normalize glutamatergic transmission in over-activated circuits of the cortico-limbic system.
Due to its distribution and its functional role at the glutamatergic synapse, mGluR2 is ideally placed to normalize highly activated synapses in affected brain regions1,2. Activation of mGluR2, which is negatively coupled to adenylate cyclase, reduces presynaptic cAMP levels and thereby glutamate release. The amygdala appears to be particularly responsive to mGluR2/3 activation3,4. In vitro activation of mGluR2 at amygdala synapses has been shown to induce a specific form of synaptic plasticity, so-called long-term depression, which maintains after washout of the agonist5,6.
Elevated limbic and in particular amygdala reactivity is found in patients with major depressive disorder but is also crucial in threat response behavior such as fear and anxiety. Similar to mGlu2/3 receptor agonists, mGluR2 PAMs have been shown to cause anxiolytic-like behavior in rodents by using various test paradigms7-9. As predicted from rodent behavior, a mGluR2/3 agonist has been shown to attenuate fear-potentiated startle in humans and to reduce generalized anxiety disorder symptoms in human proof-of-principle trials10-12. Thus, preclinical and early clinical data may indicate efficacy in threat response behavior presumably by reducing amygdala activity.
A previous Phase 2a clinical trial with a mGluR2 PAM for anxious depression failed its primary outcome measure and has been discontinued. However, treatment with JNJ-40411813 showed efficacy signals on several anxiety measures and on all depression measures (secondary outcome)13.
In summary, positive modulators of mGluR2 may limit excitatory neurotransmission at over-activated circuits known to be important for emotional control and may contribute to the potential therapeutic benefit in psychiatric indications associated with aberrant cortico-limbic activity.
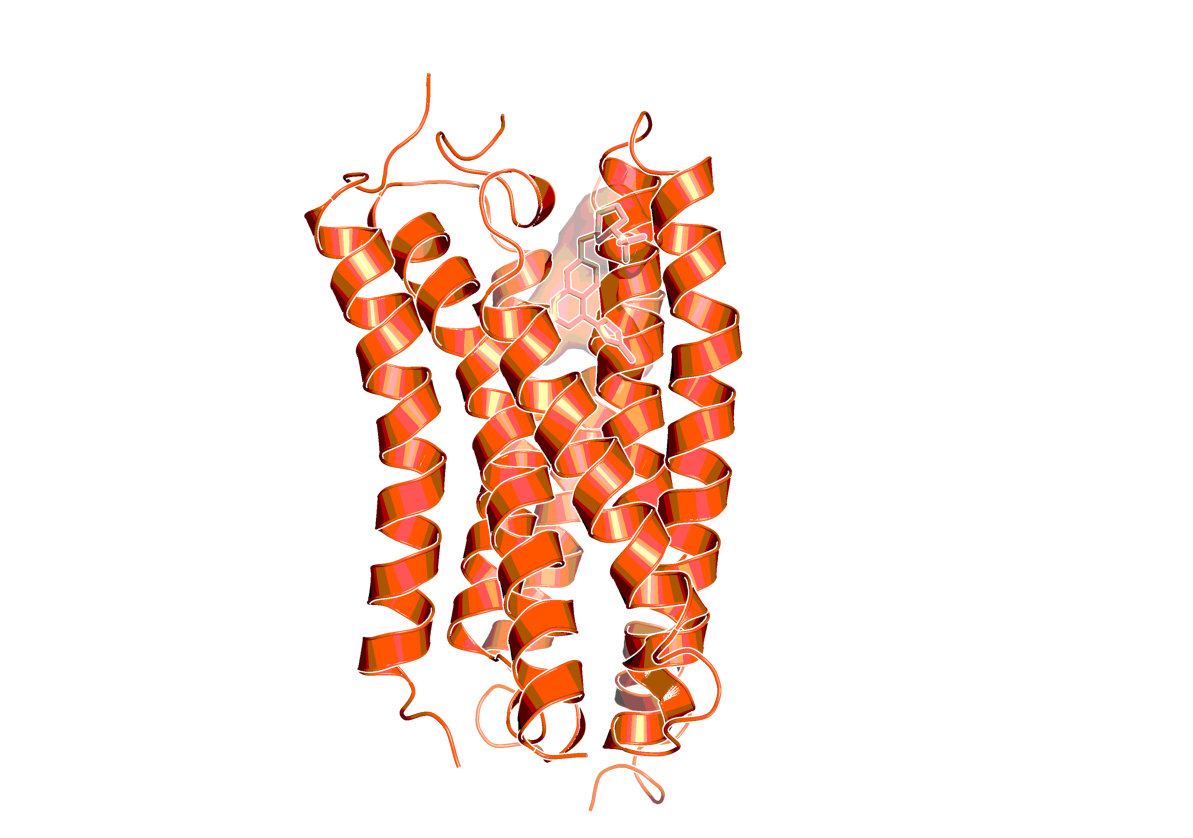
Structure of mGluR2 bound to a negative allosteric modulator (NAM563; PDB code: 7EPE14)
BI-4737 shows the following in vitro potency across species: human GTPγS EC50 = 11 nM; rat GTPγS EC50 = 3 nM.
| Probe name / Negative control | BI-4737 | BI-4576 |
| MW [Da]a | 343.47 | 318.38 |
| human GTPγS EC50 (nM)b | 11 | >10,000 |
| rat GTPγS EC50 (nM)b | 3 | - |
a The molecule is supplied in salt form; for the molecular weight of the salt, please refer to the vial label.
b Molecular activity has been determined in vitro by measuring 35S-GTPγS binding to isolated membranes from CHO cell lines expressing human or rat mGluR2. Compounds were tested in presence of glutamate at EC10 for positive modulation (EC50) of mGluR2 activity.
Membranes were suspended in 20 mM Hepes buffer (pH 7.4) containing 100 mM NaCl, 3 mM MgCl2, 1 µM or 10 µM GDP, 10 µg/ml saponin and probed at 10 µg protein per test. PAM activity was determined in presence of 1 µM glutamate. [35S]GTPγS was added to final concentration of 0.1 nM and incubated for 60 min at room temperature. Reaction was terminated by rapid filtration and filter-bound radioactivity was counted.
BI-4737 shows medium predicted clearance and a high Caco-2 efflux ratio.
| Probe name / negative Control | BI-4737 | BI-4576 |
| logD @ pH 7.4 | 2.13 | 2.5 |
| Solubility @ pH 6.8 [µg/ml] | >71 | 188 |
| MDCK permeability Pappa-b/b-a @ 1µM [*10-6 cm/s] | 53 | n.a. |
| MDCK efflux ratio | 1 | n.a. |
| Caco-2 permeability AB @ pH 7.4 [*10-6 cm/s] | n.a. | 11.8 |
| Caco-2 efflux ratio | n.a. | 0.8 |
| Microsomal stability (human/mouse/rat) [% QH] | <23 / <23 / 61 | 41 / - / 74 |
| Hepatocyte stability (human/mouse/rat) [% QH] | 26 / 46 / 57 | n.a. |
| Plasma protein binding (human/mouse/rat) [bound %] | 59 / 63 / 54 | n.a. |
| CYP 3A4 (IC50) [µM] | 1.5 | >50 |
| CYP 2C8 (IC50) [µM] | 23 | n.a. |
| CYP 2C9 (IC50) [µM] | 29 | 3.3 |
| CYP 2C19 (IC50) [µM] | 50 | n.a. |
| CYP 2D6 (IC50) [µM] | 50 | 40.5 |
| CYP 2B6 (IC50) [µM] | 2.3 | n.a. |
BI-4737 shows good exposure upon 25mg/kg p.o. dosing as well as 50mg/kg i.p. dosing (higher i.p. doses not tested). Oral dosing of 100mg/kg did not lead to significantly increased exposure compared to 25mg/kg.
| BI-4737 | MOUSEA | RATB |
| Clearance [% QH] | 51 | 125 |
| Mean residence time after i.v. dose [h] | 0.8 | 0.8 |
| tmax [h] | 0.80 | 0.5 |
| Cmax [nM] | 1,300 | 171 |
| F [%] | 79 | 24 |
| Vss [L/kg] | 2.1 | 2.1 |
a i.v. dose: 0.43 mg/kg (solution in 25% HP-b-CD, acidified with 0.1M HCl to pH6), p.o. dose: 4.3 mg/kg (suspension in Natrosol 0.5%)
b i.v. dose: 0.43 mg/kg, p.o. dose: 3 mg/kg
Receptor occupancy of BI-4737 has been investigated by ex vivo radioligand binding of a proprietary selective radioligand [3H] BI-491 to rat brain (Fig.1). The compound has been administered s.c. in the indicated dose range of 0 to 1mg/kg and brain tissue was prepared for autoradiography at 1 hour post administration. Radioligand was added at a concentration of 60nM for an incubation period of 30 min at room temperature. The binding was determined after extensive wash and receptor occupancy was calculated and plotted vs. the plasma concentration (Cplasma) from individual animals. 50% receptor occupancy has been achieved at a Cplasma of 84 nM, reflecting an IC50 of about 24 nM Cunbound, which is in range of the EC50 determined on recombinant rat mGluR2.
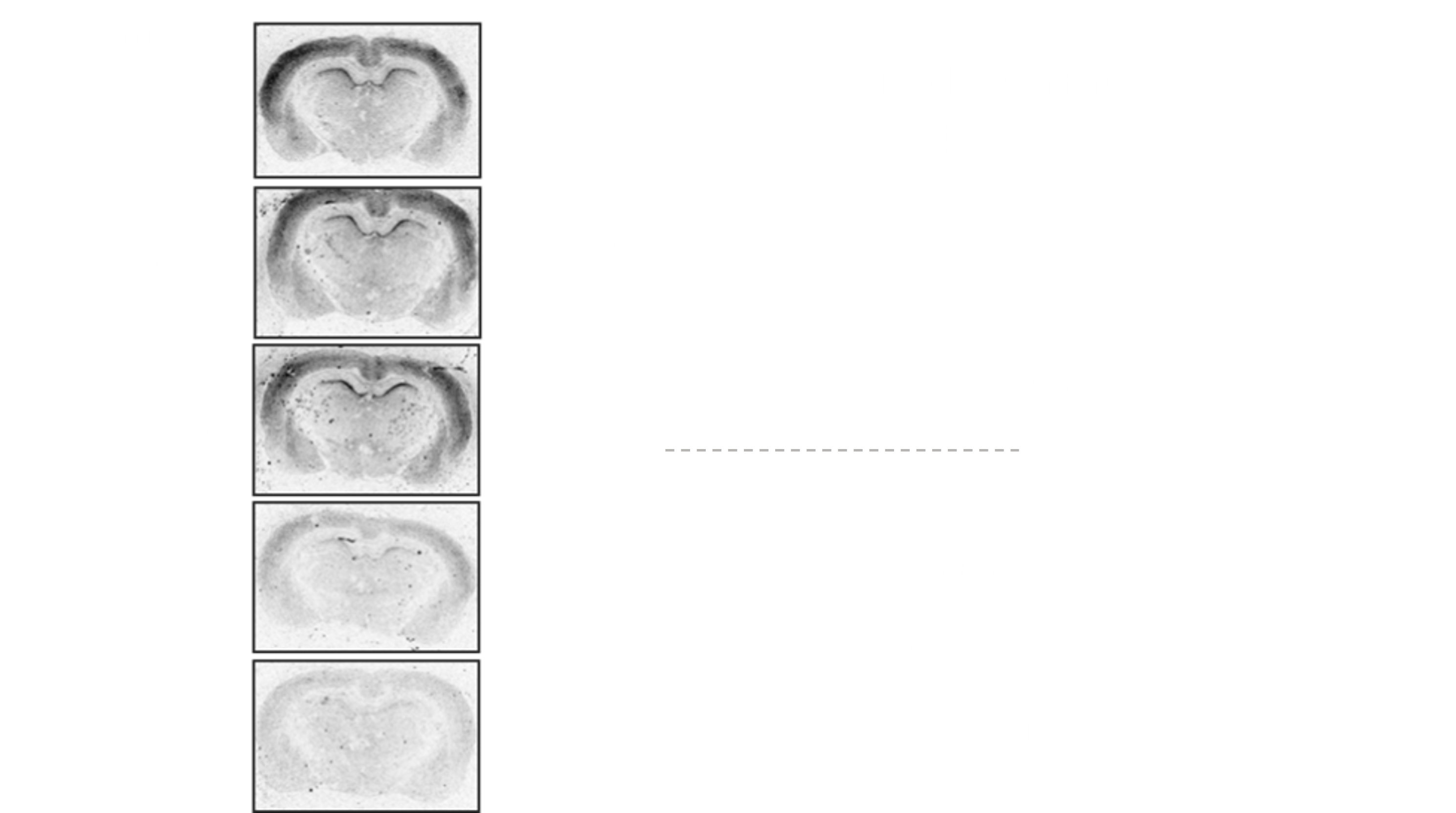
Figure 1. Receptor occupancy of BI-4737 investigated by ex vivo radioligand binding of a proprietary selective radioligand [3H] BI-491 to rat brain.
The in vivo activity of BI-4737 was determined with the rat hyper-locomotor test. BI-4737 or the reference compound LY 3547409 (mGluR2/3 agonist) were administered s.c. 30 min prior to S-ketamine with indicated doses. Experiment was performed in light and the ambulatory distance travelled was quantified for 30 min after S-ketamine administration (Fig.2).
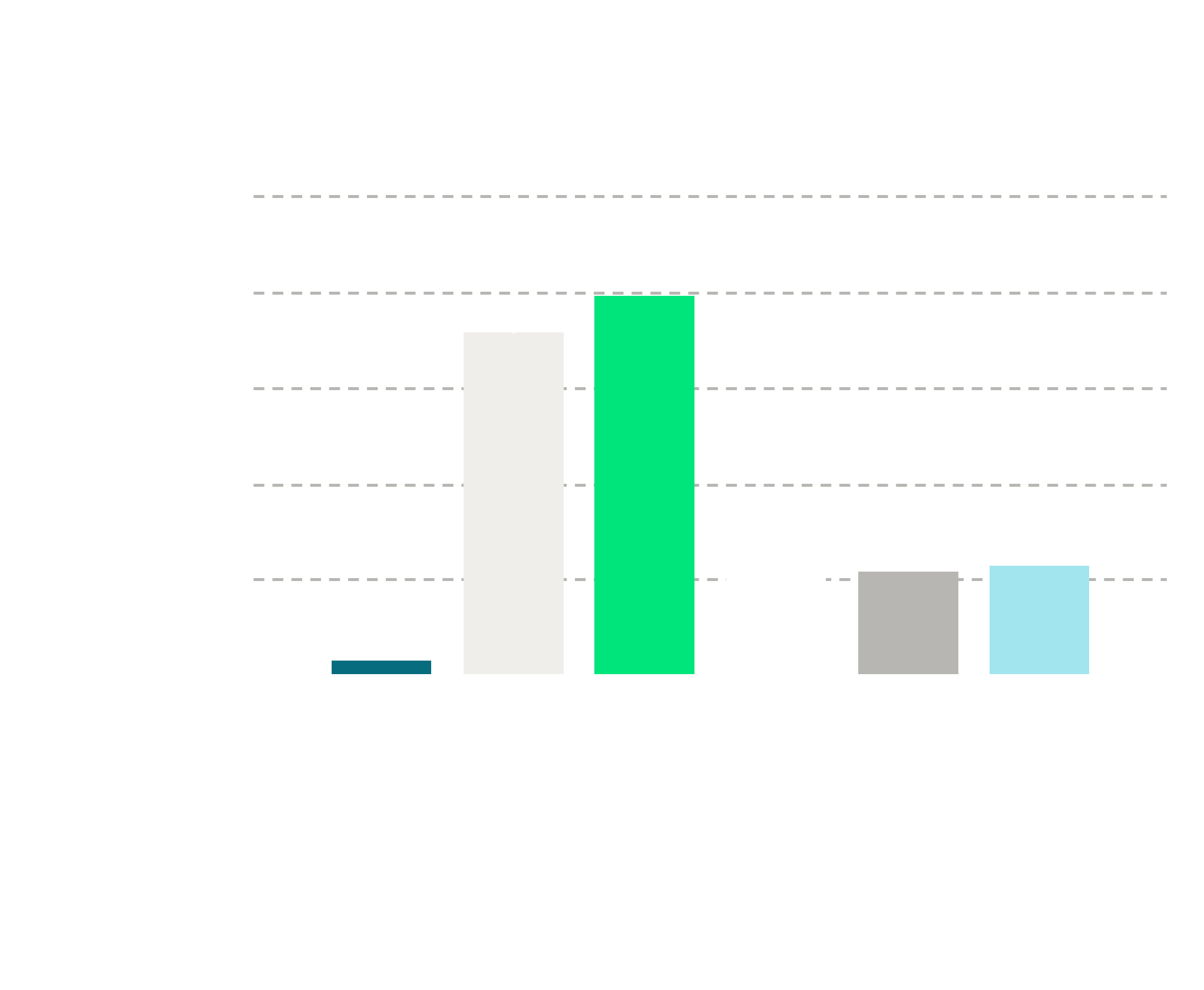
Figure 2. In vivo activity of BI-4737 monitored in the rat hyper-locomotor test. BI-4737 or the reference compound LY 354740 were administered s.c. 30 min prior to S-ketamine (10 mg/kg s.c.). Distance travelled was quantified for 30 min after ketamine administration. Statistical analysis was performed by ANOVA Dunnett's multiple comparisons test (vs. vehicle + ketamine); group size was 7-8 rats/group; ** p<0.01, *** p<0.001.
BI-4576 is a closely related structural variant of BI-4737 with a different heteroaromatic moiety that causes loss of activity. It is offered as a negative control.
BI-4576 which serves as a negative control.
BI-4737 showed 1 hit in the SafetyScreen44™ panel with >50% inhibition @ 10µM (PDE4D2). The negative control BI-4576 showed no hits in the SafetyScreen44™ panel with >50% inhibition @ 10µM.
| SELECTIVITY DATA AVILABLE | BI-4737 | BI-4576 |
SafetyScreen44™ with kind support of  | Yes | Yes |
| Invitrogen® | Yes | No |
| DiscoverX® | No | No |
| Dundee | No | No |
Download selectivity data:
BI-4737_selectivityData.xlsx
BI-4576_selectivityData.xlsx
BI-4737 is a Positive Allosteric Modulator (PAM) of the metabotropic glutamate receptor 2 (mGLuR2), a neuronal GPCR negatively coupled to adenylyl cyclase.
Antagonists reversibly reverse chemical LTD induced by group I, group II and group III metabotropic glutamate receptors
Lodge D., Tidball P., Mercier M. S., Lucas S. J., Hanna L., Ceolin L., Kritikos M., Fitzjohn S. M., Sherwood J. L., Bannister N., Volianskis A., Jane D. E., Bortolotto Z. A., Collingridge G. L.
Neuropharmacology 2013, 74, 135–146
Metabotropic glutamate 2 receptor potentiators: Receptor modulation, frequency-dependent synaptic activity, and efficacy in preclinical anxiety and psychosis model(s)
Johnson M. P., Barda D., Britton T. C., Emkey R., Hornback W. J., Jagdmann G. E., McKinzie D. L., Nisenbaum E. S., Tizzano J. P., Schoepp D. D.
Psychopharmacology 2005, 179(1), 271–283
Biphenyl-indanone A, a positive allosteric modulator of the metabotropic glutamate receptor subtype 2, has antipsychotic- and anxiolytic-like effects in mice
Galici R., Jones C. K., Hemstapat K., Nong Y., Echemendia N. G., Williams L. C., Paulis T. de, Conn P. J.
J Pharmacol Exp Ther 2006, 318(1), 173–185
In vitro and in vivo evidence for a lack of interaction with dopamine D2 receptors by the metabotropic glutamate 2/3 receptor agonists 1S,2S,5R,6S-2-aminobicyclo3.1.0hexane-2,6-bicaroxylate monohydrate (LY354740) and (-)-2-oxa-4-aminobicyclo3.1.0 Hexane-4
Fell M. J., Perry K. W., Falcone J. F., Johnson B. G., Barth V. N., Rash K. S., Lucaites V. L., Threlkeld P. G., Monn J. A., McKinzie D. L., Marek G. J., Svensson K. A., Nelson D. L.
J Pharmacol Exp Ther 2009, 331(3), 1126–1136
Efficacy and safety of an adjunctive mGlu2 receptor positive allosteric modulator to a SSRI/SNRI in anxious depression
Kent J. M., Daly E., Kezic I., Lane R., Lim P., Smedt H. de, Boer P. de, van Nueten L., Drevets W. C., Ceusters M.
Prog Neuropsychopharmacol Biol Psychiatry 2016, 67, 66–73
Crystal structure of mGlu2 bound to NAM563
Du J., Wang D., Lin S., Han S., Wu B., Zhao Q.
PDB 2021
When you plan a publication, please use the following acknowledgement:
BI-4737 was kindly provided by Boehringer Ingelheim via its open innovation platform opnMe, available at https://www.opnme.com.