PI3K gamma inhibitor
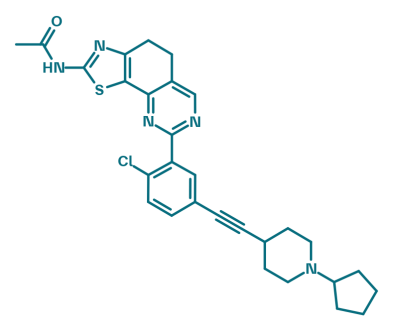
BI-9222
BI-9222 is a selective phosphoinositide-3-kinase gamma (PI3Kγ) inhibitor1. PI3Kγ is a class I phosphoinositide 3-kinase that produces PI-(3,4,5)-P3 from PI-(4,5)-P2 in response to activation of GPCRs, thereby regulating intracellular signaling pathways. It is primarily expressed and active in immune cells, controlling processes such as cell migration, respiratory burst, inflammation, and immune responses. BI-9183 can be used as the negative control2.
More information
Phosphoinositide 3-kinases (PI3Ks) are a family of lipid kinases that phosphorylate phosphatidylinositol-(4,5)-bisphosphate on the 3’ position, generating signaling molecules that regulate critical cellular processes such as growth, survival, metabolism, and immune responses. The PI3 kinase (PI3K) pathway starts with the activation of cell surface receptors like receptor tyrosine kinases (RTKs), G-protein-coupled receptors (GPCRs), or immune receptors1.
PI3Ks are categorized into three classes (Class I, II, and III)4,5, with Class I being the most extensively studied due to its role in diseases like cancer, inflammation, and autoimmune disorders. Class I PI3Ks consists of four isoforms: PI3Kα, PI3Kβ, PI3Kγ and PI3Kδ, each with distinct tissue distribution and functions.
PI3Kα and PI3Kβ are ubiquitously expressed and are involved in general cellular processes6, including proliferation and metabolism. In contrast, PI3Kγ and PI3Kδ are predominantly expressed in leukocytes, making them critical regulators of the immune system.
PI3Kγ is primarily active in innate immune cells, such as macrophages and neutrophils, where it regulates cell migration, respiratory burst, inflammation, and immune responses expressed in immune cells, including macrophages, neutrophils, and T-cells, where it governs critical functions such as immune cell activation, migration, and trafficking. Activation of PI3Kγ occurs downstream of G-protein-coupled receptors (GPCRs) through interactions with Gβγ subunits and Ras proteins, leading to the generation of phosphatidylinositol (3,4,5)-trisphosphate (PIP3), a key secondary messenger involved in cellular signaling. Dysregulation of PI3Kγ activity has been implicated in the pathogenesis of several diseases, including chronic inflammation, cardiovascular disorders, and cancer, highlighting its role as a central mediator of immune and inflammatory responses. Targeting PI3Kγ has therapeutic potential in treating inflammatory and autoimmune diseases by modulating these processes7.
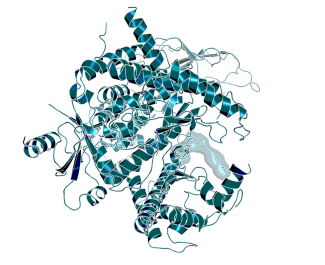
Protein Ligand Complex: X-ray structure of PI3Kγ in complex with BI-9222, structure determined at Boehringer Ingelheim
BI‑9222 is a potent and selective inhibitor of PI3kγ, showing sub‑nanomolar activity and reduced potency against the other class I PI3K isoforms.
| Probe name / Negative control | BI-9222 | BI-9183 |
| MW [Da]a | 532.1 | 401.3 |
| PI3 Kinase γ (IC50) [nM]b | 0.6 | 120 |
| PI3 Kinase δ (IC50) [nM] | 62b | 2067c |
| PI3 Kinase α (IC50) [nM]b | 273.2 | - |
| PI3 Kinase β (IC50) [nM]b | 96.1 | - |
a For the salt form you will get, please refer to the label on the vial and for the molecular weight of the salt, please refer to the FAQs
b assay conditions: assay conditions apply to all measured isoforms2
c Assay condition 8: HEK293T cells were transiently co-transfected with a NanoLuc-PIK3CD fusion vector and a PIK3R1 expression vector at a 1:9 ratio and incubated for approximately 20 h. Cells were then pre-treated with NanoBRET Tracer K-3 (150 nM) and test compounds for 2 h. NanoBRET signals were measured using a PHERAstar plate reader (BMG Labtech) equipped with luminescence filter pairs of 450 nm band-pass (donor) and 610 nm long-pass (acceptor). IC50 values were determined, and dose–response curves were generated using GraphPad Prism 8 by fitting the data with a three-parameter log(inhibitor) versus response model using the following equation: Y=Bottom + (Top-Bottom)/(1+10^((X-LogIC50))).
The in vitro DMPK and CMC data for BI‑9222 show a well‑balanced ADME profile, characterized by good hepatocyte and microsomal stability, moderate solubility, and high permeability with a low efflux ratio, supporting efficient cellular uptake.
| Probe name / Negative control | BI-9222 | BI-9183 |
| logD @ pH 2, 7.4 , 11 | 1.8, 4.4, 4.8 | 4.3, n.a., 3.3 |
| Solubility @ pH 7 [µg/mL] | 4 | <1 |
| Caco-2 permeability AB @ pH 7.4 [*10-6 cm/s] | 35.3 | 18 |
| Caco-2 efflux ratio | 0.5 | 0.5 |
| Microsomal stability (human/rat) [% QH] | 66 / 57 | <23 / 49 |
| Hepatocyte stability (human/rat) [% QH] | 20 / 43 | <4 / 70 |
| Plasma Protein Binding (human/rat) [%] | >99.8 / 98.3 | 100 / 99.9 |
| CYP 3A4 (IC50) [µM] | >10 | >50 |
| CYP 2C9 (IC50) [µM] | >10 | 30 |
| CYP 2C19 (IC50) [µM] | 4.3 | >50 |
| CYP 2D6 (IC50) [µM] | 3.7 | 25.4 |
| CYP 1A2 (IC50) [µM] | >10 | 2.6 |
The in vivo DMPK data for BI‑9222 show low clearance (12% QH) and a long mean residence time of 12 hours after intravenous dosing. BI‑9222 also demonstrates moderate oral bioavailability.
| BI-9222 | Rata |
| Clearance [% QH] | 12 |
| Mean residence time after i.v. dose [h] | 12 |
| tmax [h] | 3.7 |
| Cmax [nM] | 335 |
| F [%] | 31 |
| Vss [L/kg] | 8.1 |
a i.v. dose: 0.8 mg/kg, p.o. dose: 5.6 mg/kg
BI-9183, a structurally close analog, can be used as negative control.

BI-9183 which serves as a negative control
BI-9222 inhibited 2 out of 48 tested kinases (NEK2A, DYRK1A) by greater than 50% at a 10µM concentration and 1 (M2/H) out of 7 tested in SafetyScreen44TM by greater than 50% @ 1µM. Negative control BI-9183 inhibited 7 out of 44 tested kinases (DATRANS/HU, CCKA/H, 5HT2AH_AGO, NEUP/H, DELTA2/H, A2A/H and 5HT2B/H AG) in SafetyScreen44TM by greater than 50% @ 10µM concentration.
| SELECTIVITY DATA AVAILABLE | BI-9222 | BI-9183 |
SafetyScreen44™ with kind support of  | Yes | Yes |
| Invitrogen® | Yes | No |
| Dundee | Yes | No |
Download selectivity data:
BI-9222_selectivityData.xlsx
BI-9183_selectivityData.xlsx
BI‑9222 is a potent and selective Phosphoinositide-3-kinase gamma (PI3Kγ) inhibitor for in vitro and in vivo studies, selectively targeting GPCR‑dependent immune signaling pathways that regulate immune cell migration, inflammation, and effector functions. BI‑9183 is available as a negative control.
WO2007115929
Brandl T., Maier U., Hoffmann M., Scheuerer S., Joergensen A., Pautsch A., Breitfelder S., Grauert M., Hoenke C., Erb K., Pieper M., Pragst I.
Thiazolyl-Dihydrop-Quinazoline 2007, WO 2007115929 A1.
WO2006040279
Breitfelder S., Maier U., Brandl T., Hoenke C., Grauert M., Pautsch Al., Hoffmann M., Kalkbrenner F., Joergensen A., Schaenzle G., Peters S., Buettner F., Bauer E.
PI3-Kinasen. 2006, WO 2006040279 A1.
We would like to acknowledge Susanne Müller-Knapp and Yufeng Pan from Structural Genomics Consortium (SGC Frankfurt) for their invaluable contribution in developing the assay and generating the scientific data that supported this study.
When you plan a publication, please use the following acknowledgement:
BI-9222 was kindly provided by Boehringer Ingelheim via its open innovation platform opnMe, available at https://www.opnme.com.